- Easy and scalable enzymatic double-stranded RNA (dsRNA) removal from IVT RNA samples.
- Reduces sample dsRNA to <0.005% (LLOQ) to minimize immunogenicity in cells.
- Circumvents the need for chromatographic methods like HPLC.
- Retains single-stranded RNA (ssRNA) yields.
- Combine with modified nucleoside transcription for maximum reduction of immunogenicity.
Product is for research use only (RUO)
Product Description
The Min-Immune™ Gold dsRNA Removal Kit provides a novel enzymatic
solution for the removal of double-stranded RNA (dsRNA) contamination
present in RNA samples produced by in vitro transcription (IVT). Each 25-reaction kit will treat 1,500 μg of IVT RNA or mRNA.
The removal of dsRNA from mRNA preparations has been shown to be
essential for reducing the innate immunogenic response to the mRNA in
cells.1-4 Alternative dsRNA removal methods, such as reverse-phase HPLC,5 hydroxyapatite chromatography6 and cellulose chromatography,7
are associated with high capital costs as well as reduced final product
yields. The Min-Immune™ Gold dsRNA Removal Kit provides an easy to use,
scalable method for removing the dsRNA content of IVT or mRNA preps
without a reduction of the single-stranded RNA yield.
Uses and Label Licenses for Specific Products: See below for more information.
Product Performance
dsRNA reduction
A Min-Immune™ Gold dsRNA Removal Kit treated 1.4 kb
pseudouridine-containing RNA sample was compared to an untreated sample
(Figure 1, right panel) and dsRNA Standards (Figure 1, left panel).
Triplicates of each sample and standard were immobilized on a charged
nitrocellulose membrane, then immunoblotted with a primary
dsRNA-specific antibody and a secondary antibody conjugated to
horseradish peroxidase (HRP). Signal was detected using enhanced
chemiluminescence on a Syngene® G:Box. A regression equation was
generated using the dsRNA standard curve on the left and is used to
calculate the percent dsRNA present in both the treated and untreated
samples seen on the right side of the blot. Results show a reduction of
dsRNA content in the treated sample replicates to levels below the limit
of detection (0.005%) of this assay after treatment with the
Min-Immune™ Gold dsRNA Removal Kit.

Figure 2. dsRNA
immuno-dot blot assayed with dsRNA-specific J2 antibody detection using
horseradish peroxidase and chemiluminescent detection. 1 µg of Untreated
or Treated mRNA was blotted for each experimental sample.
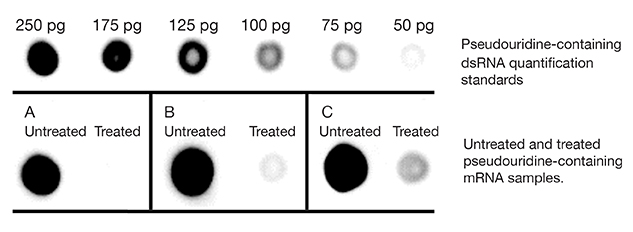
Panel A: Example result of an mRNA prep where the dsRNA content is
totally removed by treatment. This mRNA can be used for downstream
applications.
Panel B: Example result of an mRNA prep where the mRNA contains an
inherent dsRNA region which is not recognized by Min-Immune™ Gold RNase
III but is recognized by the J2 antibody. This mRNA can be used in
downstream applications.
Panel C: Example result of an mRNA prep where the dsRNA content is
not totally removed by treatment. This mRNA would require retreatment
prior to many downstream applications.
Important Store at -20°C in a freezer without a defrost cycle. Do not store at –70°C.
|
Min-Immune™ Gold dsRNA Removal Kit Contents (25 reactions) |
|
Kit Component |
Reagent Volume |
Min-Immune™ Gold RNase III (20X)
in 50% glycerol, 50 mM Tris-HCl, pH 7.5, 500 mM NaCl, 1 mM
dithiothreitol (DTT), 0.1 mM EDTA and 0.1% Triton® X-100.
|
150 μl |
|
Min-Immune™ Gold 10X RNase III Treatment Buffer |
300 μl |
ScriptGuard™ RNase Inhibitor, 40 U/μl
in 50% glycerol, 50 mM Tris-HCl, pH 7.5, 100 mM NaCl, 10 mM DTT, 0.1 mM EDTA and 0.1% Triton X-100. |
75 μl |
|
5 M Ammonium Acetate |
2 x 1.6 ml |
|
RNase-Free Water |
2 x 1.7 ml |
Materials Required, but not Supplied
- mRNA or IVT RNA for treatment.
- 70% ethanol
Optional Materials
dsRNA-specific detection system including:
- dsRNA-specific antibody (e.g., J2 antibody [Absolute Biotech-Exalpha])
- Dot/slot blotting system for use with the antibody
- An image analyzer for blot visualization and/or quantification
- dsRNA standards
References:
- Karikó, K. et al., (2011) Nucleic Acids Res. 39, e142.
- Schlee, M., Hartmann, G., (2016) Nat. Rev. Immunol. 16, 566.
- Pardi, N. et al., (2018) Nat. Rev. Drug Discov. 17, 261.
- Mu, X., Hur, S., (2021) Acc. Chem. Res. 54, 4012.
- Weissman, D. et al., (2013) Methods Mol. Biol. 969, 43.
- Kalmakoff J., Payne C.C., (1973) Anal. Biochem. 55, 26.
- Baiersdörfer M. (2019) Mol. Ther. Nucleic Acids. 15, 26.
LIMITED LABEL LICENSE
CELLSCRIPT™’s Min-Immune™ Gold dsRNA Removal Kit (“Product“)
effectively removes double-stranded RNA (dsRNA) contaminants that are
generated as by-products of making mRNA using a process comprising In vitro
transcription (IVT) of a DNA template encoding said mRNA. This Product
and the compositions and methods and uses of any thereof are covered by
United States and International Patents and Patent Applications
including and derived from World Patent Organization PCT Patent
Application Number WO 2013/102203A1 and U.S. Patent Number US12059479B2,
and further patents that issue from divisional and continuation patent
applications thereof.
By purchasing Product from CELLSCRIPT™ or an authorized distributor of CELLSCRIPT™ (herein, “Purchaser”), Purchaser receives a limited non-exclusive, non-transferable, non-sublicensable right (“Limited License”) to use the purchased Product solely for its own internal laboratory research use (the “Licensed Use”)
and Purchaser agrees to cite the name of the Product and CELLSCRIPT™ as
its source in any verbal or written public disclosure about said
Licensed Use. The Licensed Use expressly excludes any Commercial Use comprising [A] any diagnostic, prophylactic, clinical, therapeutic or other use in humans, [B] any veterinary, livestock, agricultural use in animals, and/or [C]
manufacture, distribution, importation, exportation, or sale of any
other products and/or services made using Product for which Purchaser
receives compensation of any kind, it being understood that [A]–[C] are separately and/or collectively defined as a “Commercial Use”
herein. No other license rights are granted, expressly or implied, to
make, have made, import, export, use, reverse engineer, transfer, offer
for sale, or sell Product or any other CELLSCRIPT™ product or service.
By purchase of Products, Purchaser agrees not to make, have made,
import, export, use, reverse engineer, transfer, offer for sale, or sell
Products, components of Products, or derivatives of Products, or to
provide services, information, or data obtained by the use thereof in
exchange for money or other consideration. CELLSCRIPT™ provides no
warranties (statutory or implied) concerning non-infringement of
intellectual property rights of any other parties, and all such
warranties are expressly disclaimed. This Limited License terminates
automatically if Purchaser breaches any of the terms herein.
The license remains in effect if the product is being used in
accordance with the license terms. The license is terminated immediately
upon user’s breach of any of the license terms, such as using the
product for commercial purposes without proper authorization.
If your organization is interested in using Min-Immune™ Gold
dsRNA Removal Kit or any other CELLSCRIPT™ products for a Commercial
Use, whether in catalog or custom sizes, please contact customerservice@cellscript.com with a description of your interest and needs.
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200